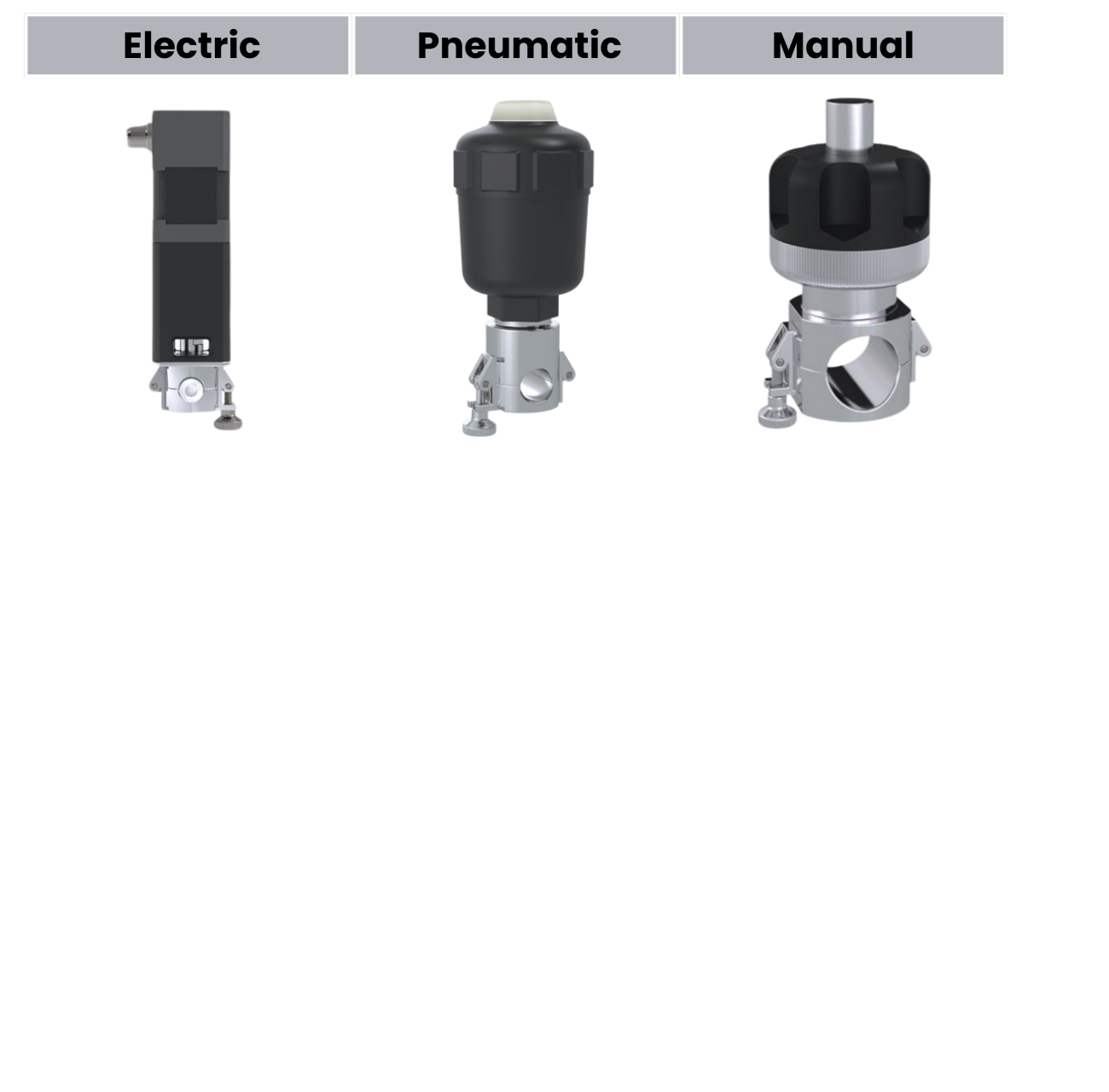
ARTēVA® Pinch Valve Series
Stainless Steel Flow Control Solutions
ARTēVA® is Alphinity’s family of reusable pinch valves, engineered for reliable, hygienic and consistent fluid control in biopharmaceutical processes.
These valves are manufactured in the United States by our stainless steel valve division, which has been designing and producing high-quality sanitary components for bioprocessing for over 30 years. Every ARTēVA® valve features a durable 316L stainless steel encapsulated pinch head built for repeated use, cleaning, autoclaving, and long-term performance.
The tubing remains fully single-use and disposable (silicone, TPE, braided or reinforced), ensuring sterility and eliminating cross-contamination risk, while the stainless steel mechanism provides uniform compression and significantly lower cost of ownership compared to fully disposable alternatives.
Core Design Advantages (All Variants)
Encapsulated 316L stainless steel pinch head — hygienic, corrosion-resistant, autoclavable
Uniform and reliable tube compression — consistent sealing even after thousands of cycles
Broad tubing compatibility — works with silicone, TPE, braided and reinforced tubing (¼″ to 1″ ID)
Tool-free tubing change-out — quick replacement of single-use tubing
Visible open/closed indication — clear operator feedback
Long service life & low maintenance — reusable design significantly reduces replacement costs
US manufactured — produced in the United States by our stainless steel valve division with over 30 years of experience in sanitary bioprocessing components
Three Actuation Options – One Reliable Pinch Design
Common Applications
Media and buffer transfer
Filtration and chromatography systems
Perfusion and cell culture feed lines
Harvest and collection paths
Viral inactivation and final fill workflows
Sampling, diversion and bypass operations
Why Choose ARTēVA®?
Consistent pinch geometry across lab-to-commercial scale — easy standardization
Premium US-made quality with over 30 years of stainless steel valve manufacturing experience
Reusable & durable — long service life and lower cost of ownership
Flexible actuation — electric precision, pneumatic speed, or manual simplicity
Hygienic and reliable — meets the highest biopharma standards for cleanability and performance
ARTēVA® Technical Data Sheets
Frequently Asked Questions- Arteva Electric
-
Arteva™ is a pinch valve system for biopharma tubing, offering manual, pneumatic, and eMaxion motorized variants. It features a central pinch point in an encapsulated 316L stainless steel body, ensuring consistent flow and closure for tubing IDs from 1/4 in. to 1 in.
-
Arteva™ revolutionizes fluid control with:
Central Pinch Design: Ensures consistent closure and flow, unlike traditional valves with inconsistent pinching.
Universal Compatibility: Works with all tubing materials (silicone, TPE including braided) from 1/4 in. to 1 in. ID.
No Cleaning Validation: Re-usable 316L stainless steel body in single-use setups.
Linear Precision (eMaxion): Delivers reliable flow response for automated processes.
Cost Savings: Reduced downtime with tool-free installation and drop-in compatibility.
-
No! Arteva™ provides drop-in compatibility with existing setups, thanks to its clamp-on design and universal tubing fit.
-
Item descSizes: 1/4 in. to 1 in. (Tubing ID)
Temperature Range: Depends on tube material (e.g., -20°C to 120°C for silicone)
Connection: Direct pinch on tube
Material: 316L Stainless Steel
Manual: Handwheel operation
Pneumatic: Air-actuated for speed
eMaxion: Bipolar stepper with encoder, 1000's of steps with micro stepping, 2 mm/s stroke, M12 connectorsription
-
Body: 316L Stainless Steel
Benefit: Hygienic, re-usable replaced as required, and compatible with all tubing types without degradation.
-
MOC Certificates
Hygiene Standards: Meets biopharma requirements
-
Yes, Arteva Electric with actuator
Encoder Feedback: Linear flow control
Visible Indicator: For all variants
M12 Connectors: Seamless integration
AI/IoT Ready: For predictive maintenance
-
Tool-Free Installation: Quick clamp-on for fast changes.
Visible Position Indicator: Reliable monitoring at a glance.
Manual Version: Hands-on control for flexibility in labs.
Pneumatic Version: Automated speed for high-volume production.
eMaxion Motorized Version: Digital precision with linear flow response for Pharma 4.0.
With eMaxion eliminate instrument air from your cleanroom
-
CGT: Sterile, low-shear control
mAbs: Precise dosing
Biosimilars: Cost-efficient flow
Continuous Manufacturing: Reliable actuation
-
Temperature: -20°C to 120°C (depending on tubing material)
Pressure: Up to 4 bar (manual/pneumatic), 6 bar (eMaxion with encoder feedback)
-
Arteva™ maintains closure reliability after 10,000+ cycles, with the central pinch design minimizing wear on tubing and valve body, ideal for continuous manufacturing (3+ days).
-
Tubing: Replace as per material lifecycle (e.g., silicone every 6 months).
eMaxion: Annual encoder calibration recommended.
-
Yes, eMaxion’s M12 connectors and 1800 micro step encoder support integration with SCADA, MES, and PLC systems. Manual and pneumatic versions can be retrofitted with sensors for basic automation.
-
Manual/Pneumatic: Max flow depends on tubing ID.
eMaxion: Linear flow up to 120% of max capacity, pressure limit 6 bar with proper tubing.
-
Validation: Includes MOC certificates, hygiene testing, and 3rd-party audits.
Sterility: Manufactured in ISO Class 7-equivalent conditions.