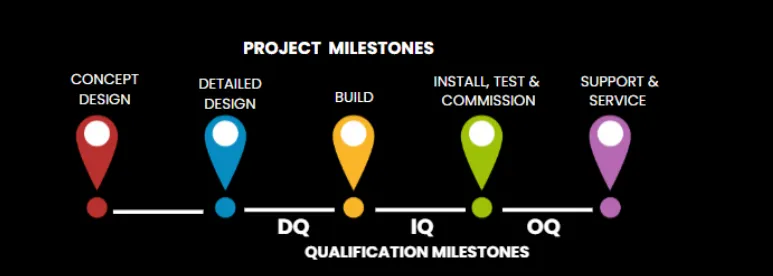
The Manufacturability Gap
The science is ready.
The equipment usually isn't.
That's the problem Alphinity was built to solve.
New therapies are failing in manufacturing, not in the lab. Fragile modalities. Viscous formulations. Unstable TMP. Scale-up failure. Alphinity designs the fluid handling systems that solve these problems at the source.